
Introduction
Medical device manufacturers face a critical procurement challenge: sourcing memory components where failures can cost lives. From MRI scanners processing terabytes of imaging data to implantable cardiac monitors, every medical technology depends on memory components that must operate flawlessly under extreme conditions. AI-enabled imaging pipelines now demand sustained data throughputs exceeding 10 GB/s, pushing memory architectures to their performance limits.
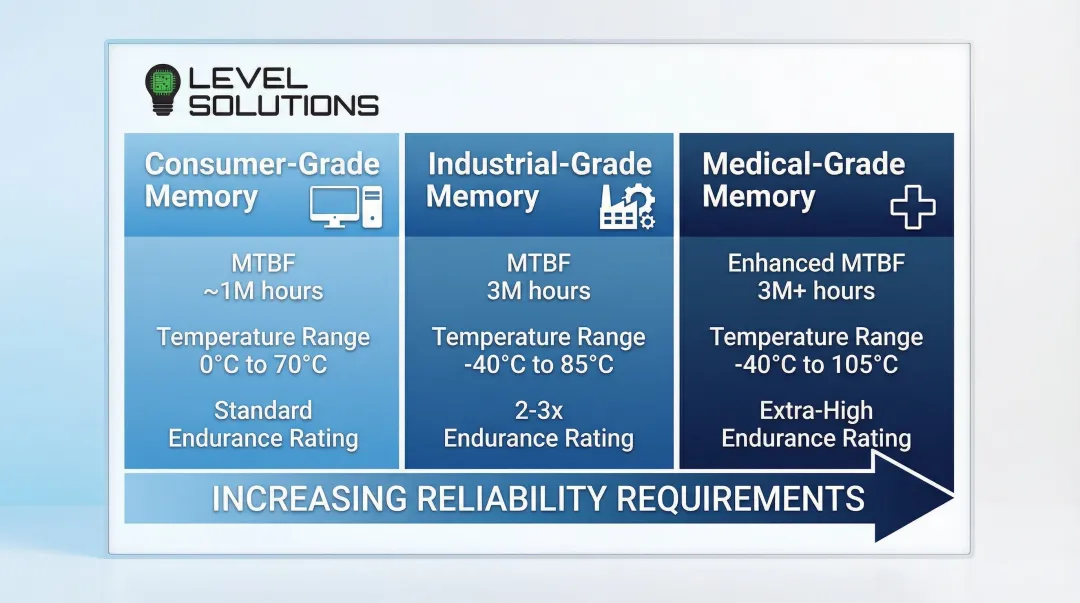
The stakes are measurably higher than consumer electronics. Industrial-grade SSDs demonstrate Mean Time Between Failures (MTBF) of up to 3 million hours compared to the typical 1 million hours of consumer counterparts.
Medical devices must also meet a complex regulatory landscape. FDA's 2026 Quality Management System Regulation (QMSR) and ISO 13485:2016 mandate rigorous component traceability and supply chain control. This article examines the specialized memory technologies, leading manufacturers, and procurement strategies that keep medical equipment reliable and compliant.
TLDR: Key Takeaways
- AI-accelerated medical imaging demands memory bandwidths exceeding 300 GB/s
- Industrial memory delivers 3x higher MTBF with -40°C to 105°C operating ranges
- Medical devices rely on DDR5 SDRAM, enterprise SSDs, and industrial eMMC with built-in protection
- Regulatory compliance requires locked BOMs, full traceability, and FIPS 140-3 Level 3 encryption
- Millions of components ship today with guaranteed long-term availability for medical device lifecycles
Why Memory is Critical in Medical Technology
Real-Time Data Processing Requirements
Diagnostic imaging equipment generates data streams that would overwhelm standard computing architectures. A single CT scan can produce over 1 GB of raw data requiring immediate processing. Meanwhile, 3D segmentation algorithms demand memory bandwidths exceeding 300 GB/s to prevent diagnostic delays.
Patient monitoring systems present different but equally demanding requirements:
- Continuous vital sign capture with zero tolerance for data loss
- Real-time processing of ECG waveforms, blood oxygen saturation, and hemodynamic parameters
- Simultaneous data logging for regulatory compliance and clinical review
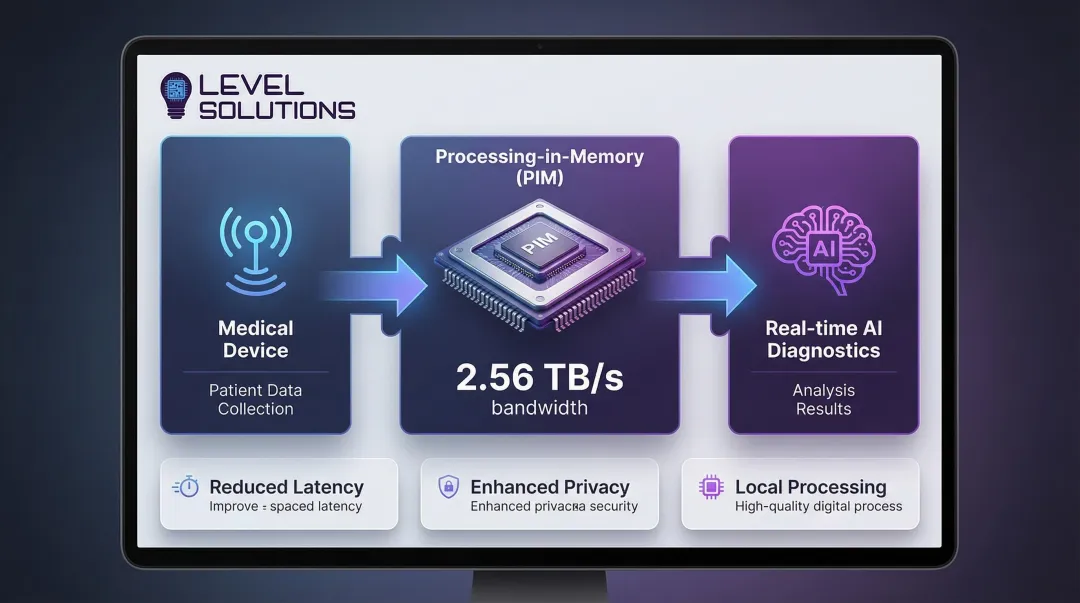
Emerging processing-in-memory (PIM) architectures demonstrate aggregate bandwidths of 2.56 TB/s for medical image processing—dramatically outperforming traditional CPU-memory configurations for data-intensive diagnostic tasks.
Reliability and Patient Safety Implications
Performance alone isn't enough—memory reliability directly impacts patient safety. A corrupted data bit in an insulin pump's dosage calculation or a failed memory cell in a ventilator's control system can have immediate, catastrophic consequences.
Medical-grade vs. consumer-grade reliability:
| Component Type | MTBF | Temperature Range | Endurance |
|---|---|---|---|
| Consumer SSD | ~1 million hours | 0°C to 70°C | Standard |
| Industrial SSD | 3 million hours | -40°C to 85°C | 2-3x lifespan |
| Medical-grade eMMC | Enhanced MTBF | -40°C to 105°C | Extra-high endurance |
Medical-grade components undergo rigorous testing protocols including extended burn-in periods, accelerated life testing, and comprehensive failure mode analysis. These rigorous validation processes identify and eliminate weak components before they reach critical medical applications.
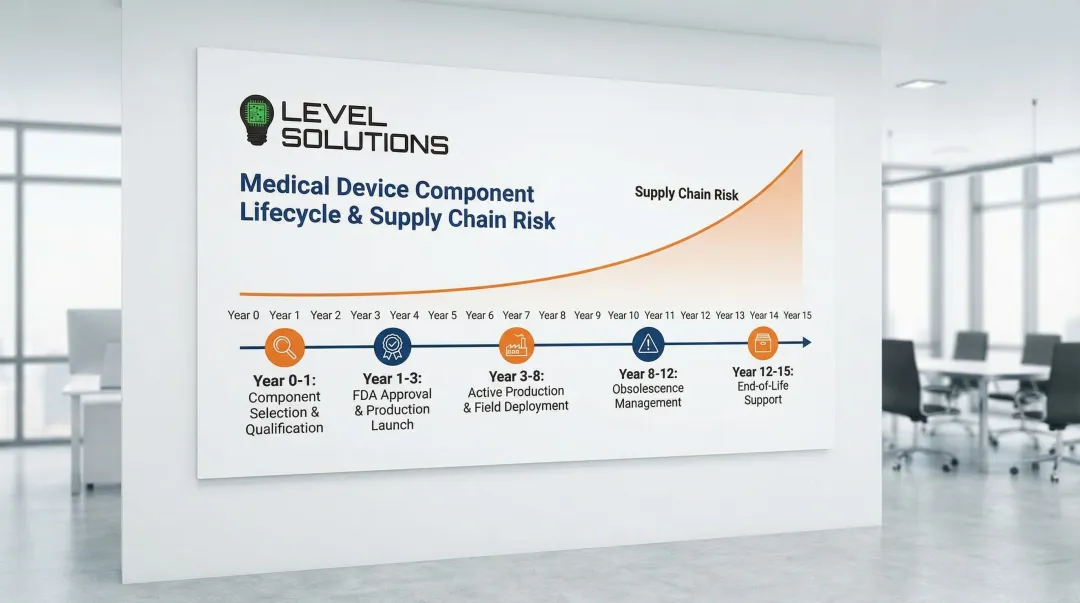
Long Product Lifecycles and Regulatory Compliance
Medical devices typically remain in service for 10-15 years, far exceeding consumer electronics lifecycles. This creates unique challenges for memory component sourcing and regulatory compliance.
Key regulatory requirements:
- FDA 21 CFR Part 820 mandates documented traceability for all components, including memory hardware and firmware
- ISO 13485:2016 requires manufacturers to evaluate suppliers based on their ability to meet quality requirements and maintain change control
- Locked BOM approach: Suppliers must guarantee no changes to critical components (controller, flash) without prior notification
Component obsolescence poses significant risks. When a memory manufacturer discontinues a specific DRAM module or flash chip, medical device manufacturers face costly requalification processes, regulatory resubmissions, and potential production halts.
Data Security and Patient Privacy
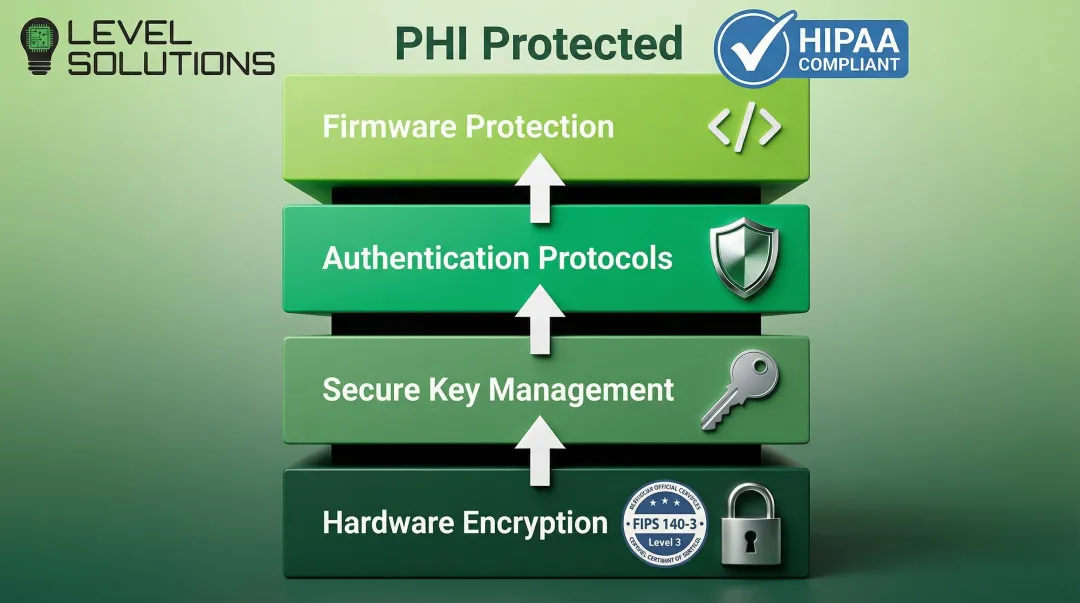
Extended product lifecycles also demand robust data security throughout the device's operational life. Protected Health Information (PHI) stored in medical device memory must be secured against unauthorized access. HIPAA compliance and FDA guidance require hardware-based encryption to protect data at rest.
Security requirements for medical memory:
- NIST-standard cryptography (FIPS 140-3 Level 3 or higher)
- Hardware-encrypted drives with self-encrypting drive (SED) capabilities
- Secure key management and authentication protocols
- Protection against firmware-level vulnerabilities
Security research has identified critical vulnerabilities in medical device memory systems, including buffer overruns and hard-coded credentials in firmware. These findings underscore the importance of secure memory implementations with validated FIPS 140-3 Level 3 encryption capabilities.
Environmental and Operational Demands
Medical memory components must operate reliably across punishing environmental conditions that consumer electronics never encounter.
Operating environment challenges:
- Temperature extremes: Operating rooms, emergency vehicles, and portable diagnostic equipment expose memory to temperatures ranging from -40°C to 105°C
- Electromagnetic interference: MRI suites, surgical electrocautery equipment, and RF-heavy hospital environments create severe EMI challenges
- Humidity and contamination: Sterilization processes, cleaning protocols, and varied clinical environments demand robust contamination resistance
- Power efficiency: Battery-operated devices require ultra-low-power memory to extend operational time between charges
ESD protection proves paramount—electrostatic discharge degrades or destroys memory components during manufacturing and field service. Medical device manufacturers require suppliers with ESD-compliant facilities and documented handling procedures.
Key Memory Technologies Powering Medical Devices
DDR SDRAM for High-Performance Computing
DDR5 SDRAM represents the current performance frontier for medical imaging workstations and diagnostic equipment. Operating at speeds from 4800 to 8800 MT/s, DDR5 provides the bandwidth necessary for real-time image reconstruction and AI-accelerated diagnostics.
Critical DDR5 features for medical applications:
- On-die ECC (Error Correction Code): Detects and corrects single-bit errors automatically, preventing data corruption in critical calculations
- Higher bandwidth: Up to 2x the data throughput of DDR4, enabling faster image processing and reduced patient scan times
- Improved power efficiency: Lower operating voltages reduce heat generation in densely packed medical equipment
DDR4 remains prevalent in established medical devices due to proven reliability and extensive validation.
LPDDR5 serves portable and handheld medical devices, offering high performance (up to 8.5 Gbps) with significantly reduced power consumption to extend battery life in point-of-care devices.
Flash Memory and eMMC for Data Storage
Embedded MultiMediaCard (eMMC) solutions deliver compact, reliable storage for patient data, device logs, and firmware in space-constrained medical devices.
Key advantages for medical applications:
- Integrated controller manages wear leveling, bad block management, and error correction
- Compact BGA package suitable for portable and implantable devices
- Industrial temperature ratings (-40°C to 105°C) for demanding environments
- Typical capacities: 4 GB to 128 GB for patient monitoring and diagnostic equipment
Industrial eMMC solutions offer 2-3x the endurance of consumer-grade components, with extra-high endurance ratings critical for continuous medical data logging applications.
NAND flash memory stores medical images, electronic health records, and device configuration data with non-volatile persistence.
SSD Solutions for Medical Imaging Archives
Enterprise solid-state drives have replaced traditional hard drives in medical imaging storage systems, offering faster access times, greater reliability, and superior shock resistance. These drives deliver essential data integrity features:
- Power Loss Protection (PLP): Integrated capacitors flush data from buffer to NAND flash during unexpected power outages, preventing data corruption
- High endurance ratings: Measured in Drive Writes Per Day (DWPD) or Total Bytes Written (TBW)—industrial NVMe SSDs offer up to 1280 TBW for continuous logging
- Enterprise-grade reliability: 3 million hour MTBF specifications ensure long-term data availability
Kioxia's CM7 and PM7 series and Samsung's PM1733 enterprise SSDs integrate power-loss protection and self-encrypting drive capabilities for medical data storage.
Specialized Memory for Embedded Medical Devices
Implantable devices, insulin pumps, and wearable health monitors require specialized low-power memory architectures. These components balance high performance with minimal power consumption:
- Low-power SRAM: High-speed cache with minimal power draw for battery-operated devices
- Pseudo-SRAM: DRAM performance with simplified SRAM-like interface
- NOR flash memory: Critical boot code and firmware storage with execute-in-place capability
- SLC NAND: Highest endurance (>100,000 program/erase cycles) for critical code storage
Remote patient monitoring and wearable health monitors use LPDDR5 or specialized ultra-low-power memory to meet strict power budgets.
Memory in Surgical Robotics and AI-Powered Diagnostics
Surgical robotics and AI-powered diagnostics require extreme memory performance for real-time processing.
High-Bandwidth Memory (HBM) and GDDR memory deliver:
- Real-time 3D visualization during robotic surgery
- AI-powered image analysis for intraoperative decision support
- Edge AI processing for faster local diagnostics without cloud latency
- Processing-in-memory architectures that reduce data movement and enable 2.56 TB/s aggregate bandwidths for advanced image processing
Redundancy and Error Correction Technologies
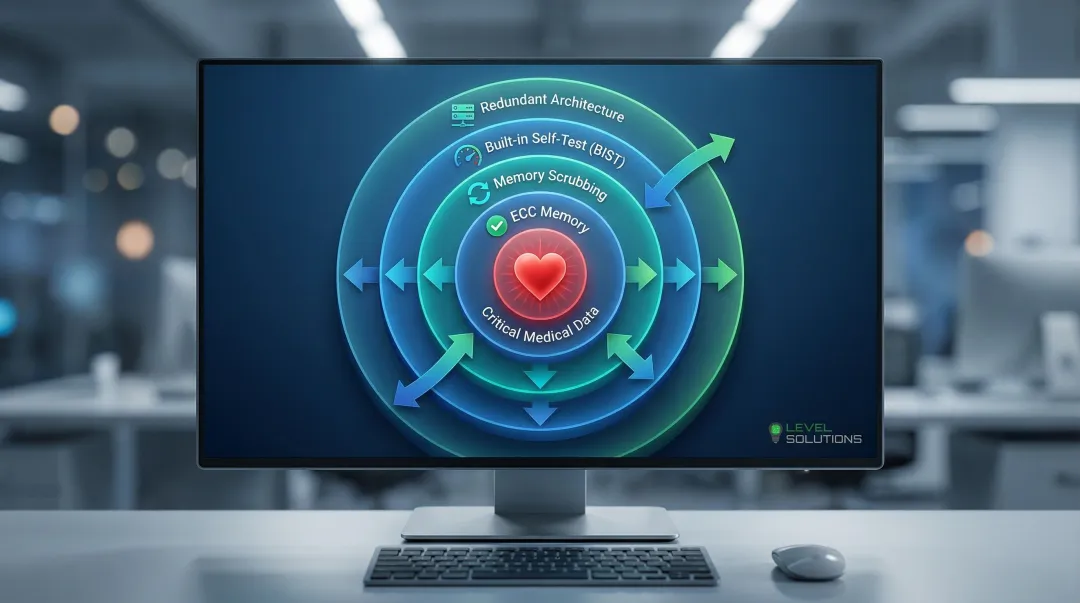
Medical equipment requires multiple layers of error detection and correction to protect patient safety.
Essential reliability features include:
- ECC memory: Detects and corrects single-bit errors, detects multi-bit errors
- Memory scrubbing: Proactively reads and corrects memory contents to prevent accumulation of soft errors
- Built-in self-test (BIST): Automated diagnostic routines verify memory integrity during startup and operation
- Redundant memory architectures: Duplicate critical data across multiple memory devices for fail-safe operation
These technologies work together to achieve the extremely low failure rates required for medical applications where memory errors could compromise patient safety.
Leading Memory Solution Providers for Medical Applications
Medical device manufacturers require memory components from vendors who understand regulatory demands, extended lifecycles, and reliability standards. The following providers offer industrial and medical-grade solutions designed specifically for healthcare applications.
Samsung Electronics
Samsung offers comprehensive medical-grade DRAM and Flash memory solutions including DDR4/DDR5 SDRAM, LPDDR5, and eMMC offerings designed for medical imaging, patient monitoring, and diagnostic equipment.
Key Samsung medical-grade offerings:
- Enterprise SSDs (PM1733 series) with power-loss protection and high endurance
- Automotive-grade SSDs rated for -40°C to 105°C operation
- DDR5 memory modules with on-die ECC for imaging workstations
- Industrial eMMC solutions for embedded medical devices
Manufacturing quality standards meet medical device certification requirements. Extended product lifecycles ensure multi-year component availability for regulated devices.
Micron Technology
Micron's portfolio spans DRAM, NAND Flash, and NOR Flash solutions specifically designed for medical and industrial applications.
Micron medical-grade highlights:
- 2100AI/AT industrial SSDs with extended temperature ranges (-40°C to 105°C)
- 3 million hour MTBF specifications for critical medical applications
- Product Longevity Program ensuring long-term component availability
- Automotive and industrial-grade memory meeting medical reliability standards
Technical support programs address lifecycle challenges unique to medical device manufacturing. Their Product Longevity Program guarantees component availability throughout regulatory approval cycles.
SK Hynix
SK Hynix provides DDR SDRAM and NAND Flash offerings for medical technology applications, emphasizing high-density, energy-efficient memory solutions. These components suit portable and implantable medical devices where power consumption directly impacts battery life.
Quality management systems comply with international standards supporting medical device regulatory requirements. However, SK Hynix markets fewer dedicated healthcare product lines compared to Samsung or Micron.
Kioxia (formerly Toshiba Memory)
Kioxia's enterprise-grade SSD and embedded Flash solutions serve medical data storage and imaging systems with advanced 3D NAND technology.
Kioxia medical-relevant features:
- CM7 and PM7 enterprise SSDs with power-loss protection
- Self-encrypting drive (SED) capabilities for HIPAA compliance
- Long-term availability commitments (up to 10 years)
- Industrial-temperature range products (-40°C to 85°C)
3D NAND technology delivers high-capacity storage with superior endurance characteristics. These features prove critical for imaging systems generating terabytes of patient data daily.

While major semiconductor manufacturers dominate medical memory markets, specialized distributors like LEVEL SOLUTIONS provide procurement expertise. Sourcing the right components requires understanding both technical specifications and regulatory timelines—expertise that bridges manufacturers and medical device OEMs.
Kingston Technology
Kingston's industrial and server memory solutions serve medical workstations and imaging systems with controlled BOM and firmware management.
Kingston medical-grade capabilities:
- Industrial SSDs with extended temperature ratings
- IronKey encrypted drives with FIPS 140-3 Level 3 validation for PHI protection
- Controlled BOM/firmware ensuring consistent performance over device lifecycles
- M.2 2280 TLC PCIe industrial NVMe SSDs with up to 1,280 TBW endurance
Specialized Industrial Memory Providers
Beyond mainstream semiconductor manufacturers, specialized vendors target medical applications exclusively. Swissbit offers healthcare-specific storage solutions with frozen BOMs and 10+ year availability guarantees.
Their designs address medical environment challenges:
- Shock and vibration resistance for portable diagnostics
- Sterilization compatibility for surgical equipment
- Hermetically sealed packages for implantable devices
- Ruggedized connectors for field medical units
Selecting Medical-Grade Memory Components: Key Considerations
Choosing the right memory components for medical devices requires balancing stringent reliability requirements with practical supply chain realities. A single component failure in diagnostic equipment or patient monitoring systems can compromise patient safety and trigger costly recalls.
Medical device engineers must evaluate candidates across four interconnected dimensions. Each plays a critical role in long-term device performance and regulatory compliance.
Reliability Metrics
Component reliability directly impacts device uptime and patient safety. Key specifications include:
- MTBF specifications (target: 2-3 million hours minimum)
- FIT (Failures In Time) rates for statistical reliability modeling
- Endurance ratings (DWPD, TBW, or P/E cycles)
- Error correction capabilities (ECC, RAID, redundancy)
Regulatory Compliance
Medical device manufacturers must navigate complex regulatory frameworks across multiple jurisdictions. Memory components need comprehensive documentation to support device certifications:
- RoHS and REACH compliance for hazardous substance restrictions
- Component-level certifications (AEC-Q100 for automotive/medical-grade)
- Supplier quality management systems (ISO 13485 compatibility)
- Documentation packages supporting FDA submissions
Supply Chain Continuity
Medical devices often remain in production for 10-15 years, far exceeding typical consumer electronics lifecycles. Component obsolescence poses significant risk:
- Component traceability and lot control capabilities
- Long-term availability commitments (10-15 year minimum)
- Change notification and obsolescence management programs
- Last-time-buy opportunities and migration support
Procurement partners like LEVEL SOLUTIONS support these requirements through comprehensive traceability systems and access to both current-generation components and legacy parts for long-lifecycle medical products.
Total Cost of Ownership
Component selection decisions extend well beyond initial purchase price:
- Initial component cost vs. lifecycle reliability
- Requalification costs if component changes occur
- Supplier stability and financial health
- Technical support quality and responsiveness
Working with Global Component Procurement Partners
Medical device manufacturers face unique supply chain challenges that specialized electronic component distributors are positioned to address.
Value of specialized procurement partners:
- Comprehensive manufacturer coverage: Access to multiple memory suppliers (Samsung, Micron, SK Hynix, and others) through a single sourcing relationship
- ESD-compliant warehousing: Proper handling and storage of sensitive memory components meeting medical device manufacturing standards
- Global sourcing capabilities: Access to allocated inventory, end-of-life stock, and alternative sourcing channels critical for long product lifecycles
Dual-Tier Availability Model
These capabilities translate directly into supply chain resilience. Level Solutions maintains millions of immediately available components in their ESD-compliant warehouse, with access to billions of additional parts from global sources.
For medical device manufacturers, this dual-tier availability model ensures supply chain continuity even during component shortages or obsolescence events. Coverage spans memory components (DRAM, SRAM, Flash, eMMC), processors, analog circuits, and passive components—enabling complete bill-of-materials sourcing with consistent quality standards and traceability documentation.
Working with experienced component procurement specialists helps medical device manufacturers navigate the complex intersection of technical requirements, regulatory compliance, and supply chain risk management that defines medical electronics sourcing.
Future Trends in Medical Memory Solutions
Ultra-Low Power Memory for Wearables and Implantables
Next-generation wearable and implantable medical devices will use emerging memory technologies offering dramatically reduced power consumption:
- FeRAM (Ferroelectric RAM): Non-volatile memory with ultra-low power consumption and high endurance
- MRAM (Magnetoresistive RAM): Instant-on capability with near-zero standby power
- Advanced LPDDR architectures: Further power reductions enabling multi-week battery life for continuous monitoring devices
These technologies will enable new categories of medical devices including long-term implantable monitors, smart bandages with embedded sensors, and ultra-compact diagnostic tools.
AI-Accelerated Edge Computing
Beyond power efficiency, these advanced memory solutions create the foundation for sophisticated on-device intelligence. Medical devices are increasingly processing data locally rather than transmitting to cloud services, driven by latency requirements and patient privacy concerns.
Emerging memory architectures for edge AI:
- Processing-in-Memory (PIM): Computation integrated directly into memory arrays, eliminating data movement bottlenecks
- Neuromorphic memory: Memory architectures mimicking biological neural networks for efficient AI inference
- High-Bandwidth Memory (HBM): Stacked memory providing massive bandwidth for AI accelerators in portable diagnostic equipment
These innovations will enable real-time AI diagnostics in handheld ultrasound probes, surgical robots, and point-of-care testing devices.
Enhanced Security Features
With more intelligence and data stored locally on medical devices, security becomes paramount. As cyber threats targeting medical devices intensify, memory security features will evolve:
- Quantum-resistant encryption: Post-quantum cryptographic algorithms integrated into memory controllers
- Hardware root of trust: Secure boot and firmware verification built into memory subsystems
- Physical unclonable functions (PUF): Unique hardware identifiers preventing counterfeiting and unauthorized access
Organizations like MDIC and JEDEC are pushing toward a unified "Medical-Class" component standard. This effort aims to formalize requirements for traceability, power-loss protection, and reliability, simplifying medical device qualification through standardized specifications.
Frequently Asked Questions
What types of memory are most commonly used in medical devices?
DDR SDRAM (DDR3/DDR4/DDR5), eMMC Flash, and enterprise SSDs dominate medical applications. High-performance imaging systems use DDR5 for real-time processing, while embedded devices rely on eMMC, and enterprise SSDs with power-loss protection serve data archives.
How does medical-grade memory differ from consumer-grade memory?
Medical-grade memory operates across extended temperature ranges (-40°C to 105°C), offers 2-3x longer lifecycles, and includes enhanced ECC with regulatory documentation. MTBF ratings reach 3 million hours versus 1 million for consumer components.
What are the key regulatory standards for memory used in medical devices?
ISO 13485 governs quality management, IEC 60601 addresses safety, and FDA 21 CFR Part 820 mandates traceability. AEC-Q100 provides component-level qualification for critical applications.
How long should medical device memory components remain available?
Medical devices operate for 10-15 years, requiring suppliers to offer long-term availability commitments and managed obsolescence programs. Leading manufacturers provide 10+ year availability for medical applications.
Which is the best company for medical equipment memory components?
Samsung, Micron, SK Hynix, and Kioxia lead medical-grade memory manufacturing. Specialized procurement partners like Level Solutions provide access to multiple manufacturers and global inventory, reducing single-source risks for long-lifecycle devices.
What role do component distributors play in medical device memory supply chains?
Specialized distributors provide global sourcing, ESD-compliant inventory management, and obsolescence mitigation. They maintain multi-manufacturer relationships, offering access to current and end-of-life components for legacy equipment support.